CRO vs Clinical Contractors: Choose the Right Trial Model
Compare CROs vs clinical contractors to protect your trial timeline. Learn coordination tax, total cost, continuity risks, and which model fits best.
RJ Gumban
2/11/20267 min read
Nothing slows a clinical trial faster than a plan that adds too much work for your team. Picking a CRO or a contractor is a big call because it changes who does the job and who owns the results.
Recent ICON research shows 55% of clinical sites take over five months to start, and 66% hit constant budget delays. These stem from regulatory, site, sponsor, or vendor factors. If handoffs or choices clog up, the schedule slips.
Before you decide, let’s break down how each model supports clinical research trial execution and what it will ask of your team.
Understanding CROs and Clinical Contractors
Ask yourself: Does your clinical trial need integrated support to reduce oversight, or targeted expertise for specific gaps? Both meet Good Clinical Practice (GCP) and regulatory requirements, but they shift workloads in unique ways.
What is a Contract Research Organization (CRO)?
A contract research organization (CRO) manages Phase I to IV clinical trial delivery as an outsourced partner, sometimes referred to as a clinical research organization when the focus is strictly on clinical trials.
With a full-service CRO, you get a team with standard processes for planning, trial design, clinical data management, tracking, and quality assurance. This reduces the time your internal team spends on daily tasks and updates.


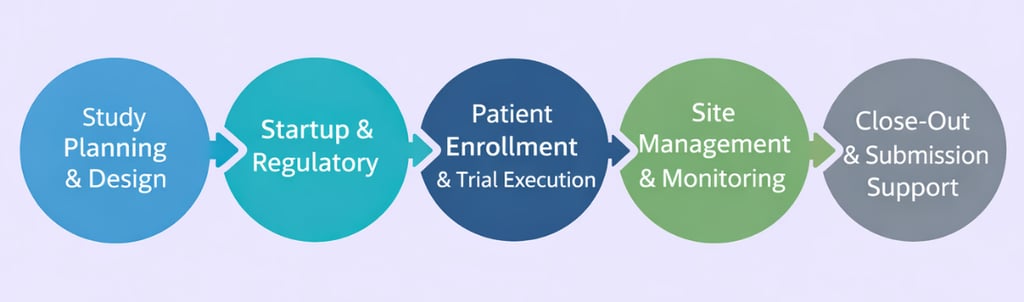
You get scale and built-in coverage through one vendor, but you’ll trade some flexibility for a more standardized way of working. Here’s what this approach typically looks like:
Value: Less sponsor coordination; structured reporting, project management, and governance.
Added Value: Risk management, liability coverage, proprietary tools for compliance.
Best fit: Phase I to IV trials across therapeutic areas where you need integrated delivery, multi-vendor coordination (laboratory services, eCOA, IRT), and support with regulatory submissions and timelines.
Cost: Higher (covers management layers, processes, and coverage).
Drawbacks: Higher upfront costs; limited customization.
3 Risks When Choosing a CRO vs Clinical Contractors
Each model changes the operational work in a clinical trial in a different way. Success depends on which risks your team can realistically handle in clinical research. Use these three lenses to weigh both CROs and contractor models without bias.
1. Miscalculating the Coordination Tax
The coordination tax is the time lost to alignment, decisions, and handoffs in clinical research. While contract research organizations usually absorb this burden, 47% of sites still rate sponsor-CRO comms as average/poor.
This disconnect creates an immediate drag at the clinical site level and can slow patient recruitment.
In contrast to CROs, the contractor model shifts that tax back to your internal clinical project manager. Managing an independent clinical research associate separately can quickly become a bottleneck for a lead who’s already stretched thin.
On the other hand, when you bring in an experienced freelance CRA with a clearly defined scope, they can often take the work and run with it with minimal oversight, which helps reduce coordination load.
What is a Clinical Contractor?
Clinical contractors are role-specific experts you plug into a clinical trial when you need extra lift, whether that’s a clinical project manager, a clinical research associate, or support for clinical data management.
They can also help unblock patient recruitment and expedite site start-up or ethics submissions when timelines are tight. They’ll work closely with the principal investigator and site staff, while your team stays in control.
You get flexibility without committing to full CRO infrastructure, while still managing handoffs and documentation. Here’s what this approach typically looks like:
Value: Rapid deployment and direct access to specialized expertise with streamlined processes.
Added Value: Cost-effective for niche tasks; retains sponsor control.
Best fit: Targeted scopes, short-term gaps, or modular tasks in therapeutic areas with stable trial designs, where an internal lead manages research outcomes without full CRO infrastructure.
Cost: Lower to variable (pay for time and expertise per role).
Drawbacks: Requires internal project management; established processes and systems, and risks inconsistent documentation or regulatory requirements.
RELATED: Uncovering the Substance of CROs
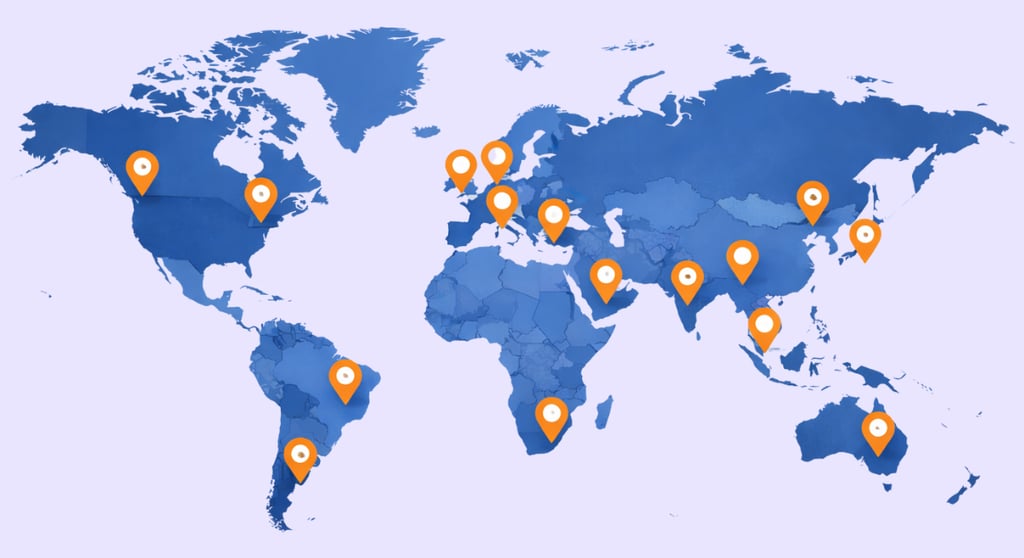
Per WCG's 2025 Clinical Research Site Challenges Report, 30% of sites cite staffing as a top challenge, with Society for Clinical Research Sites (SCRS) noting turnover up to 35-61%—friction that stalls investigational site schedules and patient recruitment around protocol design, artificial intelligence implementations, and regulatory requirements in both models.
2. Focusing on Rate Instead of Total Cost
Rates are easy to compare in a clinical trial budget, so it’s tempting to choose based on the cheapest line item. But in clinical trial ops for biotechnology and drug development platforms, “total cost” is shaped by how much structure you’re paying for and whether it actually reduces workflow friction.
On the CRO side, the risk is overbuying management layers for narrow or well-defined work. While massive clinical research organizations offer immense scale, the layers of management and approval typical of large CRO models can add coordination overhead and slow even simple decisions on narrowly scoped work.
Teams can reduce this by defining work packages tightly and setting clear decision rights and escalation paths upfront.
Applied Clinical Trials reports sponsors achieved efficiencies, including several hundred thousand dollars in savings, by shifting specific tasks from these large contract research organizations to smaller specialists.
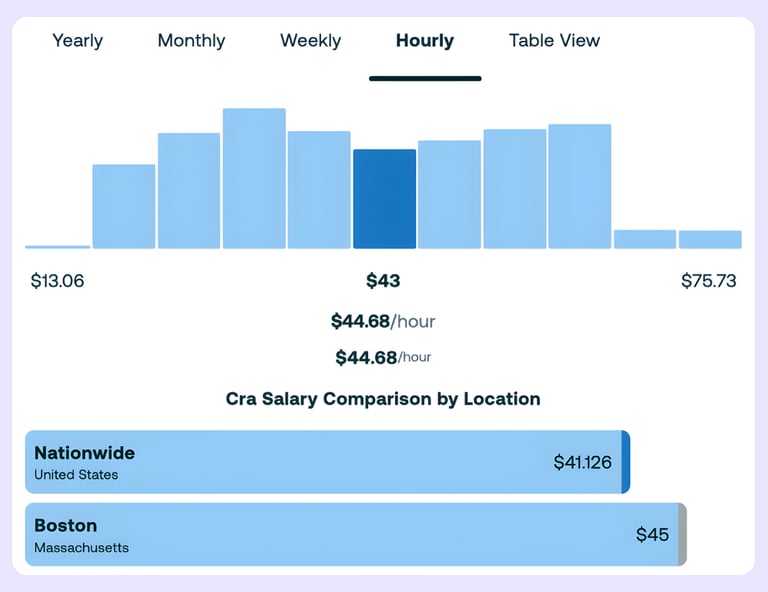
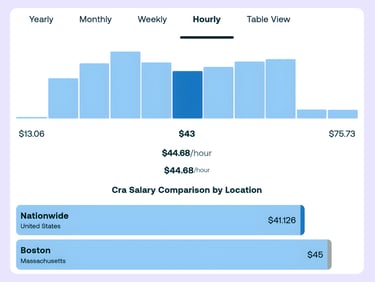
On the contractor side, the trap is undercounting the loaded cost. ZipRecruiter estimates that a clinical research associate in Boston earns about $44.68 per hour, but if you source through an agency, typical markups of 25% to 40% can push the bill rate to about $56 to $63 per hour.
When you add the time required for oversight, coordination, and onboarding, that “low” rate can rival or exceed a flat CRO fee for some programs. This is especially true when the staffing provider lacks clinical trial expertise and requires heavier sponsor-side management.
Although not all staffing models are the same. Specialized clinical staffing partners that work exclusively in clinical research, understand clinical trial workflows, and scope roles tightly can reduce sponsor-side management load and avoid unnecessary cost inflation compared to generalist agencies.
3. Ignoring Continuity and Single Points of Failure
Continuity risk exists in both models, just in different forms. Clinical research organizations usually have backup staff, but clinical trial handoffs and team changes still slow work when context and trial data aren't documented well. BDO reports that median clinical research associate (CRA) turnover hit 22% in 2024, affecting even major providers like IQVIA, and it takes 1.6 months to fill those roles.
Contractors help you skip the big vendor, but they can create a "single point of failure." If your freelance clinical research associate leaves or becomes unavailable, the project can stall without the built-in redundancy or bench depth typical of CROs, unless you have a coverage plan in place through your internal team or a specialized partner such as Freelance Clinical that can quickly source a replacement.
The fix varies by model:
For CROs, leverage service-level agreements (SLAs) and internal resources for smoother backups.
For contractors, include contract clauses for knowledge transfer, notice periods, and succession planning.
In either path, build a backup plan early and keep your handoffs simple.
Deciding Which Model Fits Your Clinical Trial
Now that we’ve covered how CROs and contractors work and the risks each model introduces, it’s time to make the choice practical. This isn’t meant to be exhaustive. It’s meant to help clinical development teams pick the setup that’s most likely to run smoothly for your team and your clinical trial.
Use the decision matrix below to sanity-check where you land across the criteria we’ve already discussed.
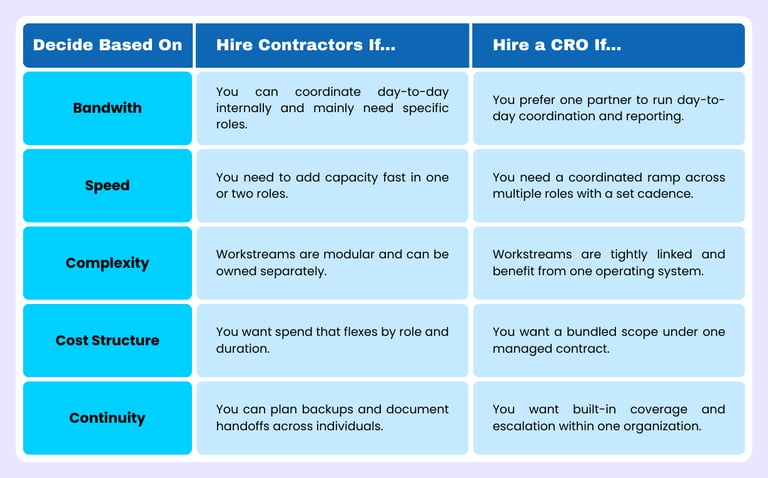
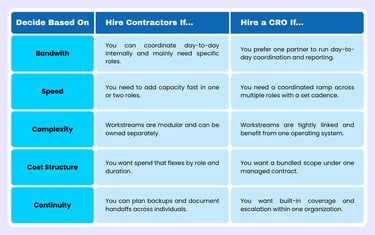
If you're still on the fence, ask yourself one question:
Can you (or your team) maintain direct oversight for the clinical trial while plugging in targeted specialists for specific roles?
Yes → A clinical contractor is often the practical fit. You keep ownership of decisions, drive study progress according to your needs, bring in role-specific expertise, and scale support as the clinical study protocol evolves.
No → A CRO is often the practical fit. You delegate ownership of day-to-day execution, rely on an integrated delivery model, and use established structure and coverage to reduce internal oversight and coordination demands.
The Decision Depends on Your Operational Reality
At the end of the day, you must decide who will drive the bus. Contract research organizations take the weight off your shoulders, while contractors give you total control as long as you can lead them. Both paths work well depending on your staff size and the risks you're willing to take.
Freelance Clinical helps you eliminate the coordination tax. We find experts in 60+ countries for pharmaceutical companies and life sciences teams across therapeutic areas, supporting outcomes research, drug development programs, and rare disease trials. That support comes with four practical benefits:
$0 Recruitment Fees: You only pay for the work you get.
48-Hour Turnaround: We move as fast as your trial does.
No Paperwork Stress: We take care of the contracts and payroll for you.
Continuity Support: If coverage changes, we help identify a backup or replacement when needed.
Start your search and build your flexible clinical trial team today!
SOLUTIONS
Disclaimer: Freelance Clinical provides clinical research staffing, vendor sourcing, and related support services. We do not identify, list, or compare all available providers. Information and recommendations are general in nature and do not constitute endorsements, guarantees, or legal, regulatory, or clinical advice. Clients must conduct their own due diligence before engaging third parties. Governed by the laws of New South Wales, Australia.
ABN: 12 671 763 643 · © 2026 Freelance Clinical Pty Ltd. All rights reserved.


COMPANY
JOIN OUR NETWORK


