The Sponsor’s Guide to Building a Flexible Clinical Trial Team
Sponsors use flexible staffing to cut delays, lower overhead, and stay in control. See how the right setup keeps clinical trials on track.
RJ Gumban
7/8/202511 min read
If you’ve ever seen timelines slip because a key role went unfilled or a vendor couldn’t keep up, you’re not alone.
63% of research sites cite staffing and retention as their top challenge.
Flexible staffing closes those gaps fast with qualified consultants, while you stay in control of quality, timelines, and cost.
This guide walks you through how to build a flexible team, bring in the right consultants, and keep your trial moving without giving up control.
What does flexible staffing mean?
Flexible staffing means hiring independent consultants to handle defined parts of a study. You bring them in quickly to meet specific needs without relying on long vendor contracts or expanding internal teams.


Key differences from traditional CRO models
CROs deliver fixed packages. You outsource a function, and the CRO assigns internal staff to manage it. If the study needs to be changed, you go back to the contract.
With flexible staffing, you stay in control. You choose the consultant, manage timelines directly, and adjust quickly as priorities shift.
Ask yourself: Are we working with timelines that are still shifting? Do we need regulatory guidance before committing to a full-service partner?
Example: You might be a sponsor preparing for a first-in-human trial, and the IND deadline is approaching fast. Your internal team is small, and the CRO hasn’t started yet. Hiring a freelance regulatory lead could help you meet your submission deadline on time and avoid a costly delay.
Mid-study staffing disruptions or scope adjustments
Clinical trials rarely go exactly as planned. A new protocol amendment, faster enrollment than expected, or a CRA leaving mid-study can create pressure that your current team may not be ready for.


Essential roles for a flexible clinical team
You can bring in freelance support across almost every function in a clinical trial. But from our experience at Freelance Clinical, these are the roles that matter most when you want to move fast, stay compliant, and stay in control.
Clinical project lead
Think of this person as your right hand. A freelance project lead keeps your timelines on track, aligns vendors, and makes sure the entire team is rowing in the same direction.
Bring one in when your trial needs stronger coordination, but you don’t want to build out an internal PM team. They’re also invaluable when language or cultural differences between sponsors, CROs, and sites begin to affect alignment.
A consultant with local experience can bridge those gaps and keep the study moving smoothly.
Clinical research associates (CRAs)
Freelance CRAs step in to handle monitoring, site visits, and local compliance, especially in regions where your CRO might not have strong coverage. They're a great option when you’re expanding into new markets or just need extra hands on the ground.
Medical monitor
A freelance medical monitor provides medical oversight throughout the trial. They review safety data, assess adverse events, and support protocol adherence.
Sponsors can engage consultant medical monitors when studies involve complex endpoints, vulnerable populations, or require specialized therapeutic knowledge.
Regulatory consultant
This expert helps you navigate submission timelines, agency interactions, and country-specific requirements. If you're preparing an IND or making major protocol changes, a freelance regulatory lead can help you avoid delays and stay compliant.
Quality and compliance expert
These consultants focus on GCP compliance, audit readiness, and quality oversight. If you're prepping for an inspection or want to strengthen vendor oversight, having someone focused purely on quality can make all the difference.
Data management and biostatistics consultant
Need to clean data fast or run an interim analysis? Freelance data experts help you get it done without long-term contracts. Sponsors usually bring them in around lock or when timelines are tight for reporting and analysis.
Medical writing expert
Strong writing supports stronger submissions. Freelance medical writers can quickly draft protocols, CSRs, or IBs with therapeutic-specific precision. Call them in when you’re up against deadlines or need clean, reviewer-ready documents.
Disclaimer
This article reflects Freelance Clinical’s experience supporting sponsors across trial phases and staffing models. It is intended to inform and guide clinical teams navigating flexible resourcing strategies. While our insights are based on industry best practices, this content does not replace formal regulatory, legal, or medical advice. Sponsors should consult appropriate experts for decisions requiring regulatory approval or legal interpretation.
Advantages of flexible staffing for clinical trials
Based on what we’ve seen at Freelance Clinical, these are the biggest advantages:
Faster deployment of talent: Avoid weeks of vendor onboarding or internal hiring delays. Consultants can start in as little as 48 hours to fill urgent gaps in monitoring, site activation, or document review.
Direct oversight and fewer delays: You work directly with the consultant. No vendor layers, no waiting for handoffs. This keeps timelines clear and reduces misalignment on scope or deliverables.
Scalable and cost-efficient: Consultants are brought in for specific needs. You can scale support up or down as priorities shift, without being locked into full-time roles or bundled service contracts.
Key takeaway: Flexible staffing gives sponsors the speed, control, and precision needed to keep trials moving without overspending.
When to choose a flexible team model
How do you know it’s time to bring in outside help? Flexible staffing is not about replacing your CRO. It’s about knowing which moments call for added support, especially when time, scope, or resources are starting to stretch thin.
Early-phase trials with uncertain timelines
If you're in early development, things are often in motion. You may be drafting your protocol while trying to figure out regulatory pathways. CROs usually need a finalized scope before getting started, which isn’t always realistic at this stage.


Ask yourself: Are we seeing gaps in monitoring coverage or delays in site follow-up? Is our CRO having trouble reallocating staff as priorities shift?
Example: Imagine your study is enrolling faster than projected in Southeast Asia, but your CRO can’t assign additional CRAs in time. Bringing in a local freelance monitor could help you stay on top of site visits and avoid backlogs in data review and source data verification (SDV).
Short-term, high-impact expertise
Some milestones only need one expert, for a short time. That person might be responsible for preparing for a regulatory inspection, delivering a CSR draft, or analyzing interim data ahead of a board meeting.


Ask yourself: Are we approaching a one-time deliverable that needs someone highly experienced? Is it more efficient to bring in a consultant than to stretch the current team?
Example: Let’s say your internal team is swamped, and you need a polished CSR within two weeks. Hiring a freelance medical writer who has done this before can get the job done without overloading your full-time team.
Specialized expertise in complex indications
If you're running a study in a rare or complex indication, the stakes are higher. You need someone who’s seen this type of protocol before and knows what hurdles to expect.
Ask yourself: Are we working in a therapeutic area that requires niche knowledge? Could a specialist help avoid early design or regulatory missteps?
Example: You may be planning a rare disease trial and are unsure how to develop a realistic recruitment plan. A freelance strategist with rare disease experience can guide your team through site selection, patient advocacy outreach, and protocol refinement.
Key takeaway: Use freelance support when speed, expertise, or flexibility is needed beyond what your current team or CRO can offer.


Key takeaway: Focus on core freelance roles that drive trial execution, quality, and compliance without long-term overhead.
How to structure your flexible team
Every sponsor’s setup looks a little different. How you structure your team depends on your internal resources, trial complexity, and how much flexibility you need. Let’s explore two common team models and walk through how to tailor your setup based on trial phase.
Full freelance team model
This approach works well if you're running lean or want to stay hands-on. You bring in consultants for each key role, then have a lead consultant or in-house project owner keep everyone aligned. It's flexible, cost-efficient, and puts you in the driver’s seat.
Ask yourself: Do we need to stay agile and avoid full CRO overhead? Do we have someone who can manage freelance consultants directly?
If that sounds like your setup, a clinical project lead can keep everything aligned and on track. Freelance Clinical can help you build a full team around them. Get in touch to explore what that could look like for your trial.
For example, if you're planning a Phase I study and don’t want to engage a CRO, your team might include:
A clinical project lead to oversee execution, offer strategic guidance, and support next steps for early-phase planning
A regulatory consultant and/or site start-up (SSU) specialist to handle IND prep, ethics approvals, and local regulatory needs
A clinical project lead or CTM to manage site communications, logistics, and vendor alignment
Regional CRAs to support site startup, monitoring, and on-the-ground oversight
A medical writer to draft the protocol, investigator brochure (IB), and informed consent forms (ICF)
This model gives you full flexibility, especially if you need to move fast or test the waters before expanding.
Hybrid model (freelance + CRO)
If you’re already working with a CRO but find there are gaps, the hybrid model can help. You keep your CRO focused on core functions while bringing in freelance experts to fill in where things need more speed, clarity, or focus.
Ask yourself: Are there parts of the trial where things are falling behind? Could short-term freelance help fix delays or add missing expertise?




Say your CRO is handling your global Phase III trial, but progress is slow in one region. You might hire:
A freelance CRA with regional experience to accelerate monitoring
A regulatory writer to support submission revisions
A medical writer to take over CSR drafting
This setup lets you stay anchored to your CRO while remaining responsive and in control of timelines.
Key takeaway: Choose a full freelance model for control and flexibility, or go hybrid if you need to boost what your CRO is already doing.
Structuring by trial phase
Once you've decided on a team model, the next step is tailoring that structure to your trial phase. Different phases come with different priorities, and having the right support at the right time makes a measurable difference.
Key takeaway: Match your team model and freelance roles to each trial phase to stay efficient and avoid delays.
Risk and quality management in flexible trials
Flexible staffing only works if quality holds up. Sponsors often ask, "How do we ensure compliance? What if a consultant misses a critical step?"
The answer is structure. With the right systems in place, freelance contributors can meet the same quality benchmarks as full-time teams. Here are three practices to help sponsors manage risk while keeping operations lean.
1. Standardize how freelancers enter your workflow
Quality starts at onboarding. Don’t assume freelancers know your expectations or systems. A lightweight onboarding package sets the tone and prevents early drift.

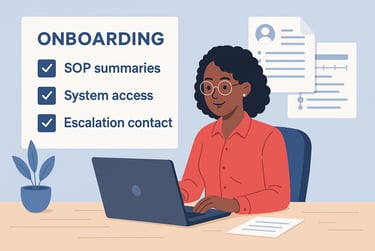
Include:
Role-specific SOP summaries covering trial goals, timelines, and handoff points
Scoped access to key trial systems (e.g., eTMF, CTMS, QMS)
Clear escalation contact, like your CTM or lead CRA
If you don’t have these systems in place yet, we can recommend our trusted vendors and help with setup to ensure your freelance team hits the ground running.
Pro tip: Before starting, freelancers should be briefed on your systems, expectations, and documentation standards. A clear onboarding flow prevents confusion and keeps timelines on track.
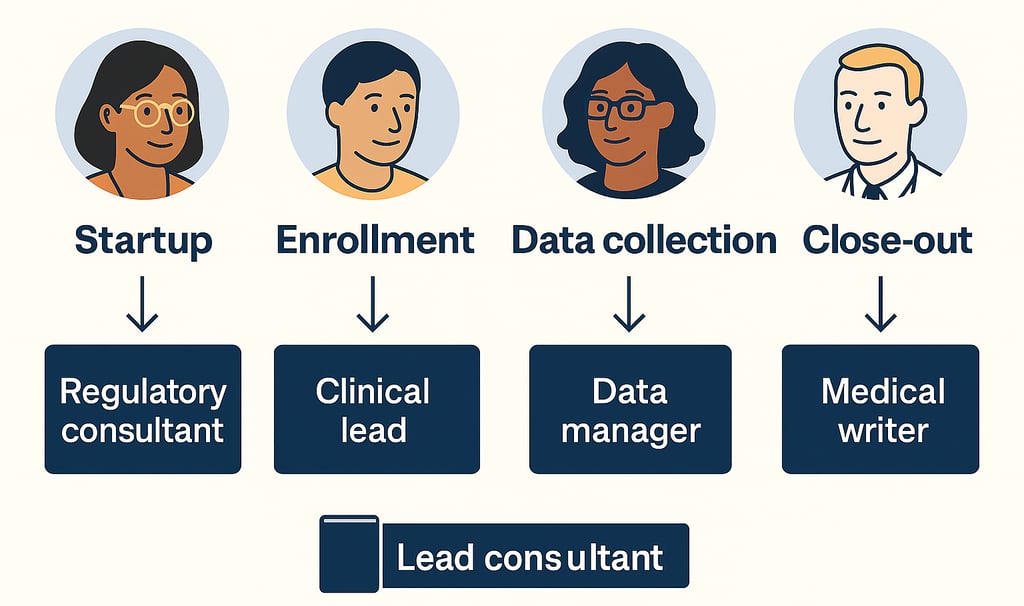
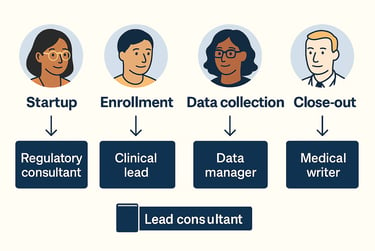
Startup: Engage a regulatory consultant and project lead to handle INDs, ethics approvals, and site setup. A startup specialist can support regional site requirements.
Enrollment: Bring in CRAs and a clinical lead to manage site relationships and keep recruitment on schedule. Their support is key when timelines start to tighten.
Data collection: Use data managers and statisticians to ensure data quality, support interim looks, and prep for database lock.
Close-out: Hire medical writers and regulatory consultants for CSRs, final submissions, and document cleanup.
Thinking ahead by phase helps you allocate freelance support strategically, so you can stay efficient without overstaffing.
2. Use milestone-based oversight, not daily micromanagement
Freelance contributors thrive on autonomy, but structure keeps quality intact. Shift from constant check-ins to outcome-focused oversight.

Build accountability by:
Tying deliverables to measurable outputs, like annotated CRFs or monitoring visit reports
Scheduling midpoint reviews to flag alignment issues early
Capturing performance feedback in trackers or CTMS notes for future reference
This creates a repeatable model for quality without slowing execution.
3. Track training and compliance like you would a CRO
Regulators don’t care who made the mistake, only that standards weren’t met. Apply the same GCP, documentation, and inspection-readiness rules across all contributors.


Ensure consistency by:
Logging GCP certificates, CVs, and SOP acknowledgments centrally (e.g., via your eTMF)
Including freelancers in audit prep and CAPA planning
Using QMS tools (like MasterControl or Qualio) to track training and version control
A lean approach is fine, but skipping these steps introduces unnecessary risk.
Key takeaway: Quality in flexible trials depends on consistent onboarding, structured oversight, and equal compliance expectations across all contributors.
How to get started with flexible staffing
You don’t need to overhaul your trial model. Getting started with Freelance Clinical is simple, fast, and designed for sponsors who need results without added complexity. Here’s how it works:
1. Tell us what you need
Fill out a short intake form. Share the role, trial phase, and how soon you need support.
2. Align with a strategist
We’ll help you scope the work, clarify what success looks like, and match you with the right consultant.
3. Start fast with full control
Get matched typically within 48 hours. You choose the consultant, manage timelines directly, and adjust support as your trial evolves.
Key takeaway: You can be matched with a qualified consultant in as little as 48 hours with no delays, no layers, and no added complexity.
Build your trial team on your terms
Trial delays don’t wait. When roles go unfilled or priorities shift, sponsors need more than another vendor. You need a partner who understands sponsor-side expectations and delivers real support fast.


Freelance Clinical connects sponsors to vetted experts who’ve led audits, submitted INDs, managed CRAs, and built systems from scratch. Our consultants are inspection-ready, sponsor-tested, and available in as little as 48 hours.
Here’s what you get on day one:
Direct access to GCP-trained, sponsor-facing professionals
Full transparency over profiles, experience, and availability
No handoffs, no delays, no layered vendor chains
Support scoped to your needs. Start with one expert or build a full team
Strategic support to help you identify the right role, fast
The process is simple. Fill out a short intake form, and our team will match you to a consultant aligned with your trial phase, systems, and quality framework.
Stop adapting to vendor constraints. Start building your trial team on your terms.
SOLUTIONS
Disclaimer: Freelance Clinical provides clinical research staffing, vendor sourcing, and related support services. We do not identify, list, or compare all available providers. Information and recommendations are general in nature and do not constitute endorsements, guarantees, or legal, regulatory, or clinical advice. Clients must conduct their own due diligence before engaging third parties. Governed by the laws of New South Wales, Australia.
ABN: 12 671 763 643 · © 2026 Freelance Clinical Pty Ltd. All rights reserved.


COMPANY
JOIN OUR NETWORK
